Curing Cancer at the Atomic Level: Designing the Future of Radioisotope Production
Harnessing a Revolutionary Therapy
A transformative medical treatment is emerging that could redefine how we fight cancer. Targeted Alpha Therapy (TAT) uses the powerful radiation emitted by decaying radioisotopes to seek out and destroy tumors with remarkable precision. Unlike traditional therapies, TAT minimizes damage to surrounding healthy tissue, offering a more focused and potentially more effective approach.
However, the radioisotopes used in TAT (such as actinium, gallium, molybdenum and lutetium) are rare and expensive to produce. As the promise of TAT becomes clearer, investment is rapidly increasing in facilities capable of sustainably and efficiently manufacturing these critical materials.
To support this precision treatment, we are leveraging our expertise in pharmaceutical manufacturing, cleanroom technology and radiological facility design to develop several state-of-the-art production centers. In collaboration with a confidential client, we are currently constructing a facility that will significantly enhance the domestic supply of medical radioisotopes in the United States. Given the use of radioactive materials, safety is paramount. Every aspect of the facility is designed to protect both personnel and the environment. Because these isotopes are intended for human injectables, the manufacturing process must meet the highest standards of quality control and regulatory compliance.
Unique Needs for a Unique Treatment
Radioisotope production facilities present a distinct set of challenges. They must accommodate highly specialized equipment, maintain strict contamination controls and meet rigorous regulatory standards while supporting complex scientific processes.
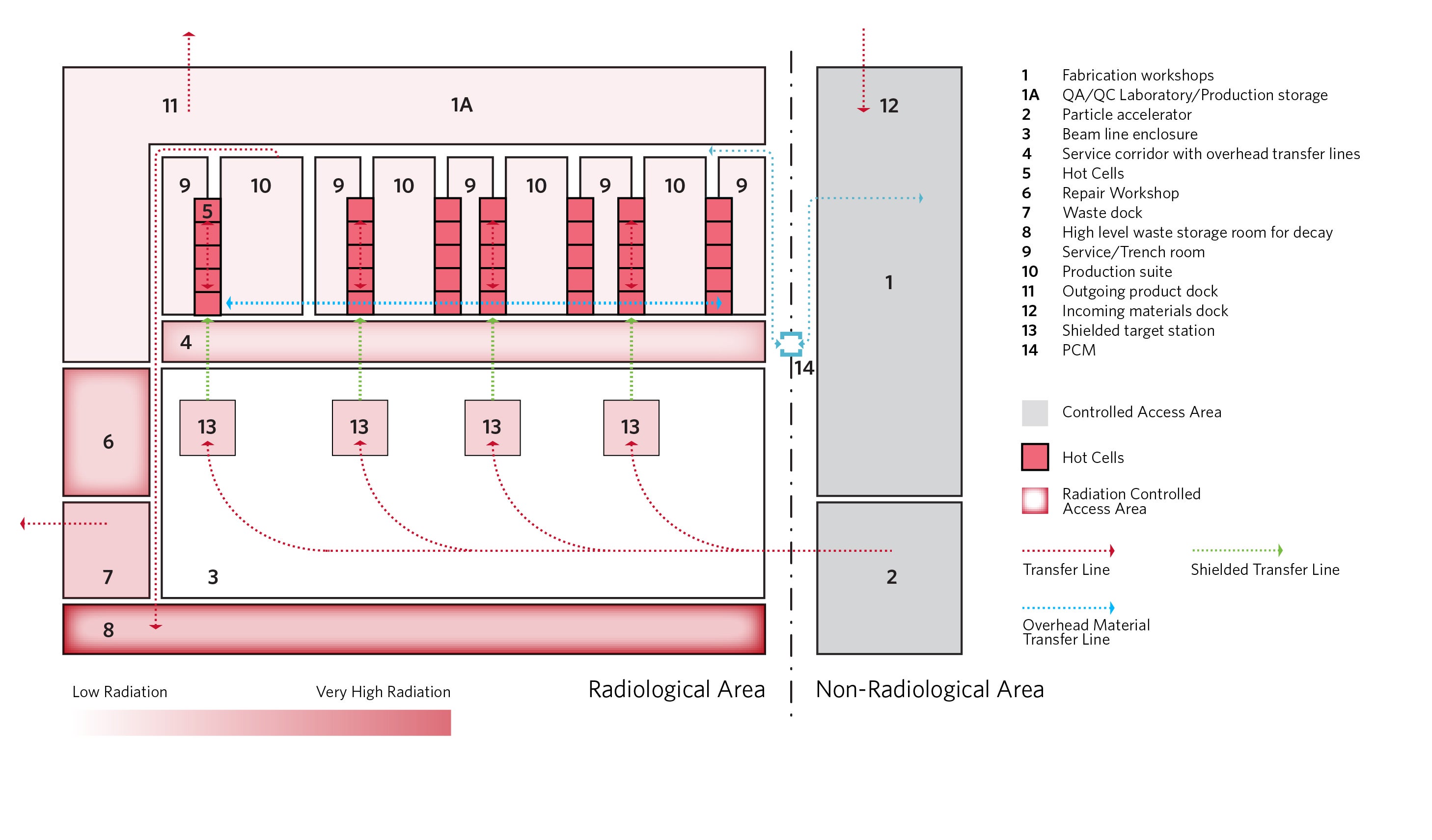
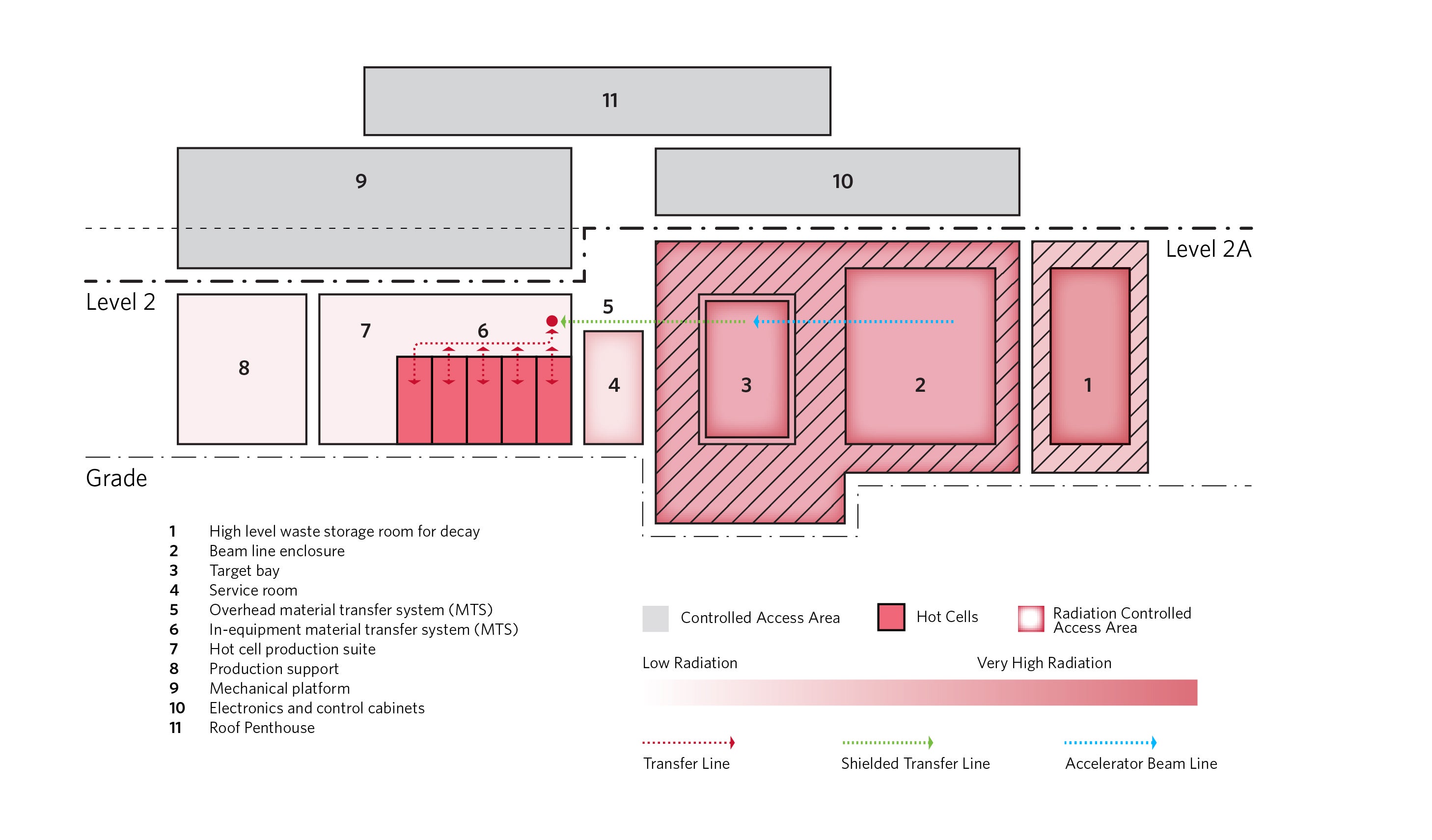
In these facilities, a linear accelerator or cyclotron bombards gaseous, solid or liquid targets with accelerated particles. These particles are guided via electromagnetic beamlines into shielded “caves,” where they interact with the targets to produce irradiated materials that emit radionuclides. Because the accelerator and beamline are exposed to high radiation levels, they are housed in heavily shielded bunkers to promote worker safety.
Once irradiated, targets are transferred using automated systems to primary containment devices for further processing. Inside hot cells—sealed, shielded enclosures equipped with robotic arms — these targets are combined with other chemical components to produce medical-grade radioisotopes. Hot cells operate under Grade A aseptic conditions and are typically arranged in modular “trains,” with each module performing a specific step in the production process.
Facilities may also include fabrication shops for producing metallic targets, metrology rooms and target development labs. Additional auxiliary spaces include quality control laboratories to test product efficacy and radioactive waste rooms to manage and potentially reuse decay waste and byproducts.
Designing for Safety at Every Level
Safety is the cornerstone of radioisotope facility design. Primary containment devices include shielded hot cells, radiological fume hoods and vented enclosures. These are reinforced by secondary containment measures such as shielded walls and ceilings to prevent contamination.
Personal protection systems are equally critical. These include radiation monitors, alarms and interlocks that restrict access or halt operations if radiation levels exceed safe thresholds. For example, accelerator systems are equipped with interlocks that prevent operation if unauthorized personnel are detected in the bunker. Technicians wear personal protective equipment appropriate to their tasks, ranging from standard lab attire to full protective suits with ventilators and oxygen support during maintenance inside hot cells.
Navigating Regulatory Requirements
Radioisotope production facilities must comply with a complex web of regulations. In the United States, the Nuclear Regulatory Commission licenses these facilities through state offices, enforcing standards based on Title 10 of the Code of Federal Regulations, which governs the management and control of radiological materials and waste. In Canada, the Canadian Nuclear Safety Commission oversees similar regulations.
Because these facilities also produce injectable pharmaceuticals, they are subject to additional oversight by the U.S. Food and Drug Administration and Health Canada, both of which enforce current Good Manufacturing Practices. Our design process integrates these regulatory frameworks from the outset to optimize facilities for long-term operational success.
Solving Complex Design Challenges
One of the most challenging aspects of designing these facilities is resolving conflicts between technical requirements. For instance, acid digestion used in radiological processes can corrode the stainless-steel liners typically found in fume hoods. Addressing this requires innovative solutions.
One approach is to modify operational procedures, such as performing acid digestion in sealed containers to minimize vapor exposure. Alternatively, engineering controls like installing fume hoods with customized liners, integrated scrubbers and washdown systems can mitigate corrosion risks. In such cases, waste must be collected in shielded containers to prevent exposure to radioactive water. These design decisions require close collaboration among architects, engineers, chemists and regulatory experts to promote safety, functionality and compliance.
Building the Future of Medicine
Radioisotope production facilities are essential to the advancement of modern medicine. As demand for targeted therapies like TAT continues to grow, so too will the need for innovative, safe and efficient infrastructure to support their development.
We are proud to be at the forefront of designing environments that enable scientific breakthroughs while protecting the people and communities they serve. Through thoughtful planning, technical excellence and a commitment to safety, we are helping to build the future of cancer treatment.